

Day 1 - Introduction to Nuclear Radiation
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
Standards-aligned
Kimette Witt
Used 19+ times
FREE Resource
19 Slides • 14 Questions
1
Day 1 - Introduction to Nuclear Radiation

2

3

4
Multiple Choice

What do these isotopes of lithium all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and electrons
protons, atomic number, and mass number
5

6

7
Multiple Choice

The photo above shows the isotopic notation for which isotope?
Carbon 12
Carbon 13
Carbon 14
Carbon 15
8
Multiple Choice
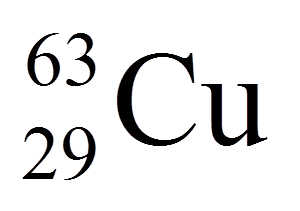
The top number in this isotope notation is
the mass number
the atomic number
the atomic mass
the neutron number
9
Multiple Choice
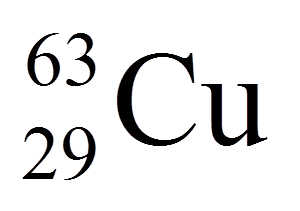
The bottom number in this isotope notation is
the mass number
the atomic number
the atomic mass
the neutron number
10

11

12

13
Multiple Choice
Which of the following is a characteristic of a nuclear reaction?
results from a change in the neutrons or protons of an element
are unaffected by pressure or temperature
cannot be sped up or slowed down
results from changing the mass number of an atom
14

15
Multiple Choice
What is an atom that has an unstable nucleus and undergoes radioactive decay?
Radiation
Radioactivity
Radioisotopes
Radium
16

17

18

19

20
Multiple Choice
X → 86Rn222 + 2He4
21
Multiple Choice
22

23

24
Multiple Choice
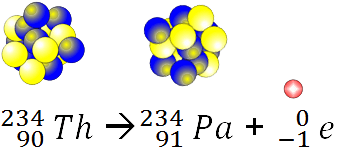
25
Multiple Choice

What was the parent element in this reaction?
23490Th
23892U
23492U
23290Th
26
Multiple Choice

What was the parent element in this reaction?
23490Th
23892U
23492U
23290Th
27
Multiple Choice
Which of the following correctly describes the charge of beta particles and alpha particles?
Alpha particles are positively charged, and beta particles are negatively charged.
Alpha particles and beta particles are both negatively charged.
Alpha particles are negatively charged, and beta particles are positively charged.
Alpha particles and beta particles are both positively charged
28

29

30

31
Fill in the Blank

What is missing from this reaction?
32
Multiple Choice
33

Day 1 - Introduction to Nuclear Radiation

Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

29 questions
Isotopes & Radioactivity
Lesson
•
9th - 12th Grade

23 questions
Stock System - Naming Ionic Compounds
Lesson
•
10th - 12th Grade

24 questions
Metric System
Lesson
•
9th - 12th Grade

29 questions
Interpreting Heating Curves
Lesson
•
9th - 12th Grade

28 questions
Ionic & Acids Naming & Formulas
Lesson
•
9th - 12th Grade

30 questions
Oxidation and Chemical Formulas
Lesson
•
9th - 12th Grade

26 questions
Properties of Life LESSON
Lesson
•
10th - 12th Grade

25 questions
Exploring Exponential Models
Lesson
•
10th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

13 questions
SMS Cafeteria Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

12 questions
SMS Restroom Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

10 questions
Pi Day Trivia!
Quiz
•
6th - 9th Grade
Discover more resources for Chemistry

13 questions
Solubility Curves
Quiz
•
10th Grade

10 questions
Exploring Nuclear Fission and Fusion
Interactive video
•
6th - 10th Grade

5 questions
Exit Check 4.1 - Balancing Chemical Equations
Quiz
•
10th Grade

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

8 questions
Labeling a Reaction
Quiz
•
9th - 12th Grade

92 questions
Chem Quarter 3 Review
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

22 questions
Swilley's Chemistry Counting Atoms
Quiz
•
9th - 12th Grade