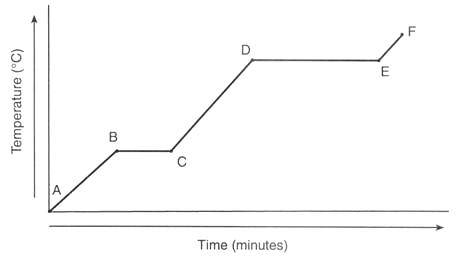
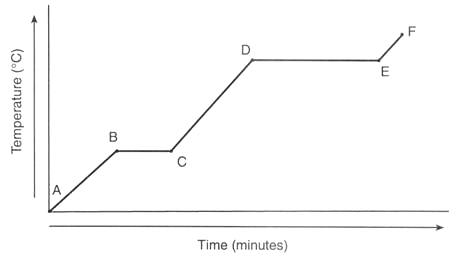
Interpreting Heating Curves
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+2
Standards-aligned
Amy Dahl
Used 54+ times
FREE Resource
11 Slides • 18 Questions
1
2
3
4
5
6
7
8
9
10
11
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
Show answer
Auto Play
Slide 1 / 29
SLIDE
Similar Resources on Wayground

20 questions
Electron Configuration
Lesson
•
9th - 12th Grade

21 questions
Le Chatlier's Principle
Lesson
•
10th - 12th Grade

19 questions
Structure of Atom
Lesson
•
9th - 12th Grade

22 questions
Verbo IR (to go)
Lesson
•
9th - 12th Grade

22 questions
Effective Communication
Lesson
•
9th - 12th Grade

22 questions
Population Data Lesson
Lesson
•
9th - 12th Grade

22 questions
Notes on Imperfect Tense AR,ER,IR and 3 Irregular Verbs
Lesson
•
9th - 12th Grade

22 questions
RiverWatch Lesson #1
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

19 questions
Stoichiometry, Limiting Reactants, and Percent Yield
Quiz
•
10th Grade

20 questions
Stoichiometry Practice
Quiz
•
12th Grade

10 questions
Formative 3BD: Ionic Bonds
Quiz
•
9th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

10 questions
Identifying types of reactions
Quiz
•
9th - 12th Grade