
Understanding Molecular Polarity
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Hard

Jennifer Brown
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What determines if a molecule is polar?
The even distribution of electrons
The speed of electron movement
The number of atoms in the molecule
The number of protons in the molecule
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
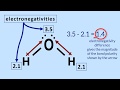
Why is water considered a polar molecule?
Because of the uneven distribution of electrons
Because it has a symmetrical shape
Because it has more protons than electrons
Because oxygen is less electronegative than hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the shape of a sulfur difluoride molecule?
Bent
Trigonal planar
Linear
Tetrahedral
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the shape of carbon dioxide affect its polarity?
It makes the molecule non-linear
It increases the electronegativity of carbon
It causes the bond polarities to cancel out
It makes the molecule polar
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when one fluorine in BF3 is replaced with chlorine?
The bond polarities no longer cancel out
The molecule becomes non-polar
The bond polarities remain the same
The molecule becomes symmetrical
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in determining molecular polarity using the flowchart?
Check if the molecule is symmetrical
Determine if any bonds are polar
Identify the central atom
Calculate the total number of electrons
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the polarity of HCl?
Neutral
Symmetrical
Polar
Non-polar
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
