
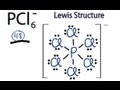
PCl6- Ion Structure and Properties
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many total valence electrons are present in the PCL6- ion?
42
48
36
54
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atom is placed at the center of the PCL6- Lewis structure?
Nitrogen
Oxygen
Chlorine
Phosphorus
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons are used to form chemical bonds in the PCL6- structure?
12
10
16
14
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of completing octets for chlorine atoms in the PCL6- structure?
It stabilizes the molecule.
It increases the molecule's reactivity.
It changes the molecule's charge.
It decreases the molecule's size.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can phosphorus have more than eight valence electrons in the PCL6- structure?
It is in Period 4 of the periodic table.
It is in Period 3 of the periodic table.
It is in Period 2 of the periodic table.
It is in Period 1 of the periodic table.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What notation is used to indicate that PCL6- is an ion?
Brackets with a negative sign outside
A positive sign outside the brackets
No special notation is needed
A negative sign inside the brackets
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
