
Understanding the CH3+ Ion
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
5 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
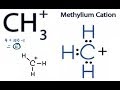
How many total valence electrons are present in the CH3+ ion?
4
5
6
7
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the best way to arrange the atoms in the CH3+ Lewis structure?
Carbon and hydrogens in a triangular shape
All atoms in a straight line
Hydrogens in the center with carbon around them
Carbon in the center with hydrogens around it
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the plus sign next to the carbon atom in CH3+ indicate?
It has gained an electron
It is neutral
It has a negative charge
It has a positive charge
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formal charge of the carbon atom in CH3+?
0
+1
-1
+2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is CH3+ considered to be unstable?
It has too many electrons
It is a very common molecule
It lacks a full octet for carbon
It has a negative charge
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
