
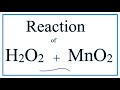
Catalysts and Decomposition of Hydrogen Peroxide
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Jackson Turner
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the products formed when hydrogen peroxide decomposes?
Hydrogen and water
Hydrogen and oxygen
Water and oxygen
Water and carbon dioxide
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is observed when hydrogen peroxide is applied to a wound?
It evaporates quickly
It produces bubbles of oxygen
It forms a solid layer
It turns blue
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of a catalyst in a chemical reaction?
It is consumed in the reaction
It slows down the reaction
It speeds up the reaction without being consumed
It changes the products of the reaction
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which enzyme in the blood acts as a catalyst for the decomposition of hydrogen peroxide?
Lipase
Protease
Catalase
Amylase
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is MnO2 written above the reaction arrow in the decomposition of H2O2?
Because it is a reactant
Because it is a product
Because it is a catalyst and remains unchanged
Because it is an inhibitor
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to MnO2 after the decomposition of hydrogen peroxide?
It is converted into oxygen
It is converted into water
It is consumed in the reaction
It remains unchanged
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
