
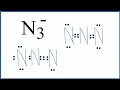
Lewis Structures and N3- Ion
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of valence electrons in the N3- ion?
14
17
15
16
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How are the nitrogen atoms arranged in the initial Lewis structure of N3-?
In a triangle
In a square
In a row
In a circle
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of moving electrons from the outside to the inside in the Lewis structure?
To change the element
To decrease the total valence electrons
To form double or triple bonds
To increase the number of atoms
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a valid alternative Lewis structure for N3-?
Two single bonds
Three single bonds
One triple bond and one single bond
Two triple bonds
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formal charge on the central nitrogen in the alternative Lewis structure with a triple bond?
0
+1
-1
-2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the Lewis structure with two double bonds considered better?
It has formal charges closer to zero
It is more symmetrical
It has more atoms
It uses fewer electrons
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total charge on the N3- ion?
-2
+1
0
-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
