
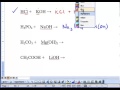
Neutralization Reactions in Chemistry
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Medium
Patricia Brown
Used 3+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a salt in the context of chemistry?
A type of base
A type of acid
A class of ionic compounds
A type of sugar
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the products of a neutralization reaction?
Metal and non-metal
Acid and base
Hydrogen and oxygen
Salt and water
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a neutralization reaction?
Oxygen and hydrogen
Hydrochloric acid and sodium hydroxide
Carbon dioxide and water
Sulfuric acid and aluminum
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to balance chemical equations?
To increase the temperature
To make the reaction faster
To ensure the conservation of mass
To change the products
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of writing water as HO in balancing equations?
To change the reaction
To make balancing easier
To simplify the equation
To increase the reaction rate
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of combining HCl and K in a neutralization reaction?
KOH and HCl
H2 and Cl2
H2O and KCl
KCl and water
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the reaction between H3PO4 and NaOH, what is the salt formed?
NaOH
NaCl
Na2SO4
Na3PO4
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
