
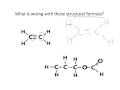
Understanding Structural Formulas and HONC Rule
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of the HONC 1234 rule?
To determine the number of bonds formed by metals
To ensure the correct number of bonds in organic molecules
To calculate the molecular weight of compounds
To identify the color of chemical compounds
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the HONC rule, how many bonds does nitrogen typically form?
Four bonds
Three bonds
Two bonds
One bond
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does oxygen form two bonds according to the HONC rule?
It needs two more electrons to fill its valence shell
It is a diatomic molecule
It has four valence electrons
It has two lone pairs of electrons
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a structural formula?
A formula that predicts chemical reactions
A diagram showing how atoms are joined together
A formula that shows the molecular weight
A list of elements in a compound
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How can you correct a structural formula that violates the HONC rule?
Add more atoms
Change single bonds to double bonds
Remove all hydrogen atoms
Increase the number of lone pairs
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a condensed structural formula?
A formula that shows every atom and bond
A simplified representation omitting some atoms
A formula that lists all elements alphabetically
A formula that only shows carbon atoms
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a condensed structural formula, what is typically omitted?
All atoms
All bonds
Carbons and most hydrogens
Only carbon atoms
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
