
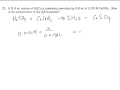
Titration Calculations and Concepts
Interactive Video
•
Chemistry, Mathematics, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in a titration calculation?
Measure the temperature of the solution
Determine the color change of the indicator
Write and balance the chemical equation
Calculate the pH of the solution
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a titration involving NaOH and HCl, what is the product formed besides water?
Sodium chloride
Sodium acetate
Sodium hydroxide
Hydrochloric acid
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert milliliters to liters in a titration calculation?
Multiply by 1000
Divide by 1000
Subtract 1000
Add 1000
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molarity of a solution if 0.01 moles of solute is dissolved in 0.038 liters of solution?
0.26 M
0.32 M
0.29 M
0.35 M
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the titration of NaOH with acetic acid, what is the salt formed?
Sodium sulfate
Sodium bicarbonate
Sodium chloride
Sodium acetate
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the stoichiometric ratio of acetic acid to sodium hydroxide in their titration?
2:1
1:2
1:1
3:1
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the titration of sulfuric acid with calcium hydroxide, what is the balanced formula for the salt formed?
CaCl2
CaCO3
Ca(NO3)2
CaSO4
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
