
Solubility and Saturation Concepts
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is one factor that can change the rate at which a solute dissolves in a solvent?
The color of the solute
The temperature of the solvent
The shape of the container
The time of day
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is solubility defined in terms of saturation?
The maximum amount of solute that can dissolve in a solvent
The ability to dissolve any amount of solute
The color change when a solute dissolves
The speed at which a solute dissolves
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
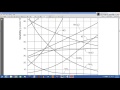
What does Reference Table G primarily show?
The density of different solutes
The solubility of different solutes at various temperatures
The pH levels of various solutions
The boiling points of various liquids
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does temperature generally affect the solubility of a solute?
Lower temperatures increase solubility
Higher temperatures decrease solubility
Higher temperatures increase solubility
Temperature has no effect on solubility
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is an unsaturated solution?
A solution that can still dissolve more solute
A solution that has no solute
A solution that cannot dissolve any more solute
A solution that has dissolved the maximum amount of solute
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
At 25 degrees Celsius, what is the solubility of NaCl in water according to the discussion?
38 grams per 100 grams of water
25 grams per 100 grams of water
50 grams per 100 grams of water
10 grams per 100 grams of water
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the solubility of potassium nitrate at 50 degrees Celsius?
100 grams per 100 grams of water
80 grams per 100 grams of water
60 grams per 100 grams of water
120 grams per 100 grams of water
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
