
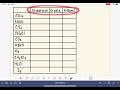
Intermolecular Forces and Boiling Points
Interactive Video
•
Chemistry, Science, Biology
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is NOT an intermolecular force discussed in the video?
Hydrogen bonding
Dipole-dipole forces
Ionic bonding
Dispersion forces
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a key requirement for a molecule to exhibit hydrogen bonding?
Presence of chlorine atoms
Presence of sulfur atoms
Presence of hydrogen and either oxygen, nitrogen, or fluorine
Presence of carbon atoms
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which molecule is likely to have dispersion forces?
Only polar molecules
Only nonpolar molecules
All molecules
Only ionic compounds
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How can you determine if a molecule has dipole-dipole forces?
By checking if it is polar
By checking if it is ionic
By checking if it is nonpolar
By checking if it has hydrogen bonding
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relationship between hydrogen bonding and dipole-dipole forces?
Hydrogen bonding occurs only in nonpolar molecules
Hydrogen bonding is weaker than dipole-dipole forces
Hydrogen bonding is a type of dipole-dipole force
Hydrogen bonding does not involve dipole-dipole forces
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which factor is NOT considered when ranking molecules by boiling point?
Intermolecular forces
Molecular weight
Color of the molecule
Presence of hydrogen bonding
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary factor that determines the boiling point of a molecule?
The number of atoms in the molecule
The shape of the molecule
The intermolecular forces present
The color of the molecule
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
