
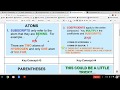
Chemical Formulas and Atom Counting
Interactive Video
•
Chemistry, Mathematics, Science
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a subscript in a chemical formula indicate?
The type of chemical bond
The number of atoms of the element
The charge of the compound
The number of molecules
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does a coefficient affect a chemical equation?
It affects the charge of the compound
It alters the chemical properties
It changes the type of elements present
It indicates the number of molecules
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the effect of parentheses in a chemical formula?
They indicate a chemical reaction
They change the state of matter
They show the charge of the compound
They group atoms to be multiplied by a subscript
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the formula Ca3(PO4)2, how many phosphorus atoms are present?
6
3
1
2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many oxygen atoms are in the compound Al2(SO4)3?
16
12
8
4
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of atoms in one molecule of Pb(NO3)2?
10
12
11
9
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If a compound has a coefficient of 3, how many times should you count the atoms in the formula?
Three times
Once
Four times
Twice
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
