
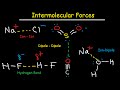
Intermolecular Forces and Properties
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Easy
Aiden Montgomery
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which factor increases the strength of ion-ion interactions?
Decreasing the size of ions
Increasing the charge of ions
Increasing the distance between ions
Decreasing the charge of ions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of interaction occurs between a sodium ion and water?
Hydrogen bond
Ion-dipole interaction
Dipole-dipole interaction
Ion-ion interaction
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a characteristic of hydrogen bonds?
They involve hydrogen and highly electronegative atoms like N, O, or F.
They are weaker than London dispersion forces.
They occur between non-polar molecules.
They are a type of ion-ion interaction.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary force present in non-polar molecules?
London dispersion forces
Hydrogen bonds
Dipole-dipole interactions
Ion-ion interactions
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which intermolecular force is the weakest?
London dispersion force
Hydrogen bond
Dipole-dipole interaction
Ion-ion interaction
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of intermolecular force is predominant in methane (CH4)?
London dispersion force
Hydrogen bond
Dipole-dipole interaction
Ion-ion interaction
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which compound is likely to have a higher boiling point, CH3OH or CH4?
CH4, because it is non-polar
CH3OH, because it is non-polar
CH3OH, because it has hydrogen bonds
CH4, because it has more electrons
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
