
Molecular Geometry and VSEPR Theory
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
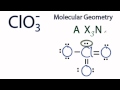
What is the main objective when analyzing the chlorate ion (ClO3-)?
To determine its color
To measure its mass
To calculate its density
To find its molecular geometry
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many oxygen atoms are present in the Lewis structure of ClO3-?
Three
Two
One
Four
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many lone pairs of electrons are present in the ClO3- ion?
One
Two
Zero
Three
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to VSEPR theory, what causes the atoms and lone pairs to spread out in ClO3-?
Nuclear attraction
Gravitational pull
Magnetic forces
Electron pair repulsion
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which theory is used to predict the shape of the ClO3- ion?
VSEPR theory
Quantum theory
Atomic theory
Relativity theory
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In AXN notation, what does 'A' represent?
The bond angle
The central atom
The number of bonded atoms
The number of lone pairs
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the 'X' in AXN notation stand for?
The central atom
The bond angle
The number of lone pairs
The number of atoms bonded to the central atom
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
