Lewis Structures with VSEPR
Authored by Lisa Thompson
Science
11th Grade
NGSS covered
Used 4+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
25 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
According to VSEPR, molecules adjust their shapes to keep which of the following as far away as possible?
Pairs of valence electrons
Inner shell electrons
Mobile Electrons
Electrons closest to the nucleus
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
How many electrons are shared in each bonding line?
1
2
3
4
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

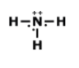
How many total valence electrons are participating in bonding in the molecule above?
8
4
2
3
Tags
NGSS.HS-PS1-2
4.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
According to VSEPR theory, what is the molecular geometry of a molecule with a central atom that has two bonding pairs and two lone pairs of electrons?
Linear
Bent
Tetrahedral
Trigonal Planar
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Which of the following is an acceptable Lewis structure for CH3Cl?
Option A
Option B
Option C
Option D
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Which of the following is the correct Lewis structure for the compound PBr3?
structure A
structure B
structure C
structure D
7.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which of the following molecules has a trigonal pyramidal shape according to VSEPR theory?
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?