
Acid-Base Titration Concepts
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Mia Campbell
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
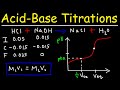
What is the balanced chemical equation for the reaction between sulfuric acid and sodium hydroxide?
H2SO4 + 2NaOH → NaHSO4 + 2H2O
H2SO4 + 2NaOH → Na2SO4 + 2H2O
H2SO4 + NaOH → NaHSO4 + H2O
H2SO4 + NaOH → Na2SO4 + H2O
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How can the concentration of H2SO4 be calculated using the M1V1 = M2V2 formula?
By using the volume of NaOH only
By ignoring the molar ratio between H2SO4 and NaOH
By considering the 1:2 molar ratio between H2SO4 and NaOH
By using the volume of H2SO4 only
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a monoprotic acid?
An acid that can donate three protons
An acid that cannot donate any protons
An acid that can donate one proton
An acid that can donate two protons
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a strong acid-strong base titration, what is the pH at the equivalence point?
Greater than 7
Less than 7
Exactly 7
Depends on the concentration
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the pH curve of a strong base-strong acid titration differ from that of a strong acid-strong base titration?
It starts at a lower pH and ends at a higher pH
It starts at a higher pH and ends at a lower pH
It starts and ends at the same pH
It remains constant throughout
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
For a weak acid-strong base titration, what is the pH at the equivalence point?
Depends on the base
Exactly 7
Less than 7
Greater than 7
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the buffer region in a titration curve?
It shows where the pH changes drastically
It indicates the start of the titration
It represents a region where pH remains relatively constant
It marks the end of the titration
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
