
Electron Configuration Review
Chemistry
10th - 12th Grade
NGSS covered
Used 10+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
27 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
This "rule" of Quantum Chemistry states that it is impossible to know both the position and velocity of an electron at the same time.
2.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
Which shape represents the shape of an "s" orbital?
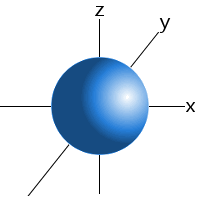
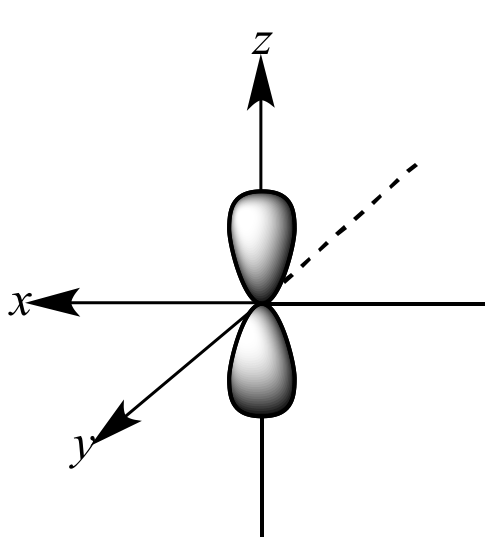
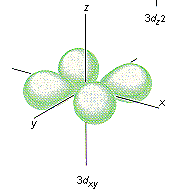
Tags
NGSS.HS-PS1-1
3.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
Which shape represents the shape of an "p" orbital?



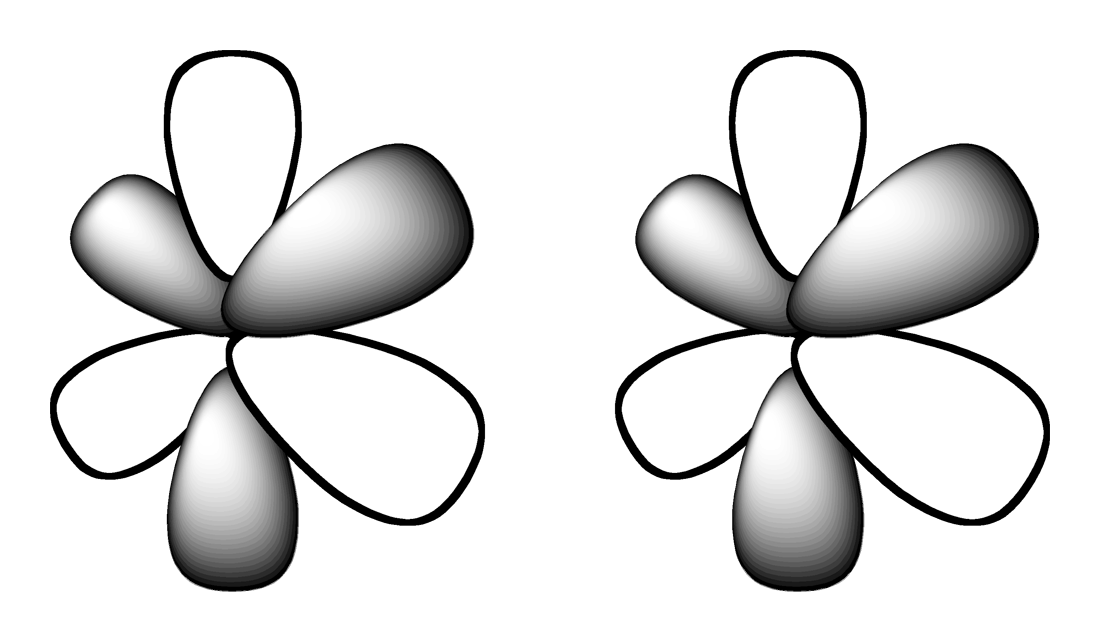
4.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
Which shape represents the shape of an "d" orbital?



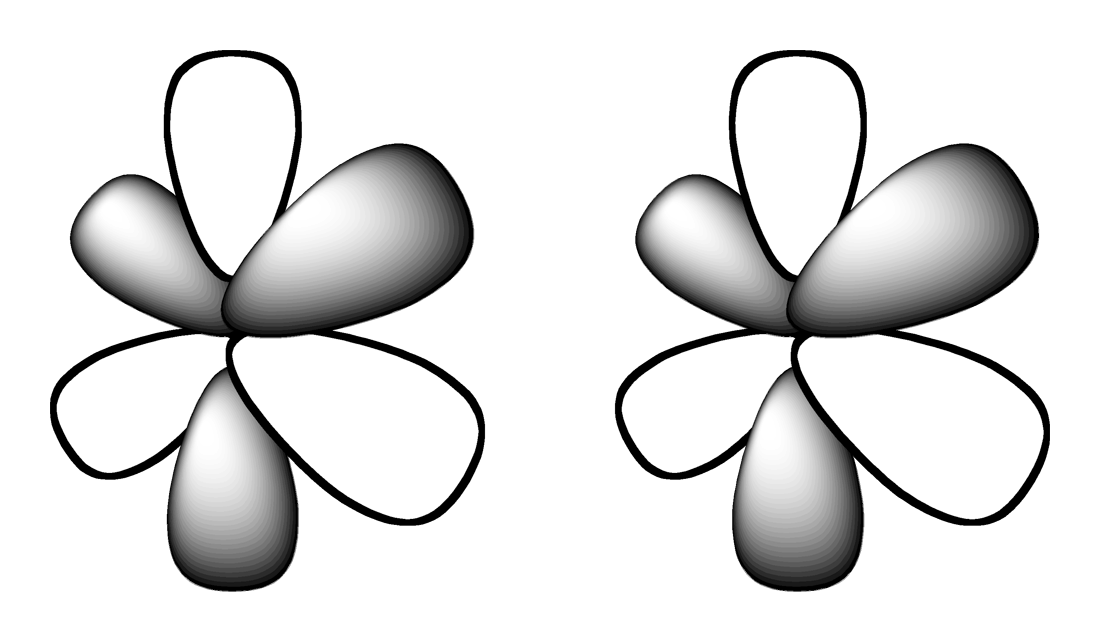
5.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
How many electrons can a single orbital hold?
1
2
3
5
6.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt

How many electrons can the p sublevel hold?
Tags
NGSS.HS-PS1-1
7.
MULTIPLE SELECT QUESTION
5 mins • 1 pt

Which of the following Aufbau diagrams are incorrect? Check all that apply.
A
B
C
D
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?



