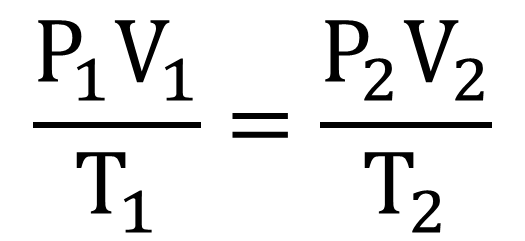Understanding the Combined Gas Law
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Genia Harris
Used 11+ times
FREE Resource
9 Slides • 11 Questions
1
2
3
Multiple Choice
4
Multiple Choice
5
Dropdown
6
7
8
Multiple Select
9
Multiple Select
10
Open Ended
11
Fill in the Blank
12
Fill in the Blank
13
14
15
Multiple Select
16
17
Multiple Choice
18
Multiple Choice
19
20
Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

16 questions
Counting Atoms
Lesson
•
9th - 12th Grade

16 questions
Stoichiometry - Limiting Reagents & Percent Yield
Lesson
•
9th - 12th Grade

18 questions
Physical Vs. Chemical Changes/Properties
Lesson
•
10th - 12th Grade

12 questions
Scientific Method
Lesson
•
9th - 12th Grade

15 questions
Senderos 2 verbos como gustar
Lesson
•
9th - 12th Grade

15 questions
Exponential vs. Linear: Growth and Decay
Lesson
•
9th - 12th Grade

15 questions
How to support claims in your writing
Lesson
•
9th - 12th Grade

15 questions
Loans intro
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

13 questions
Solubility Curves
Quiz
•
10th Grade

20 questions
momentum and impulse
Quiz
•
9th - 12th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

20 questions
Naming Covalent Compounds
Quiz
•
11th Grade

35 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade