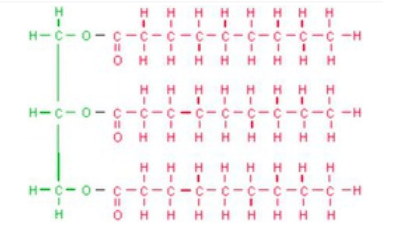
High School Organic Chemistry
Presentation
•
Science
•
10th Grade
•
Hard
Joseph Anderson
FREE Resource
69 Slides • 50 Questions
1
2
3
4
5
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
12
13
Multiple Choice
14
Multiple Choice
15
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
20
21
22
23
24
25
Multiple Choice
26
27
Multiple Choice
28
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
32
33
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
38
39
40
41
42
Multiple Choice
43
44
Fill in the Blank
45
46
Multiple Choice
47
48
Multiple Choice
49
50
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
57
58
59
60
61
Multiple Choice
62
Open Ended
63
64
65
66
67
68
69
70
71
72
73
74
75
76
Multiple Choice
77
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Fill in the Blank
83
Multiple Choice
84
Open Ended
85
86
87
88
89
90
91
92
Multiple Choice
93
Multiple Choice
94
Multiple Choice
95
96
97
98
99
100
101
Multiple Choice
102
Multiple Choice
103
Multiple Choice
104
105
106
107
108
109
110
111
112
Multiple Choice
113
Multiple Choice
114
Multiple Choice
115
116
Multiple Choice
117
Multiple Choice
118
Multiple Choice
119
Multiple Choice
Show answer
Auto Play
Slide 1 / 119
SLIDE
Similar Resources on Wayground

117 questions
Phy Science Final Exam Review
Lesson
•
9th - 12th Grade

111 questions
FInal Exam Study Guide
Lesson
•
11th - 12th Grade

105 questions
Human Circulatory System
Lesson
•
9th - 12th Grade

108 questions
APHG - Chapter 9
Lesson
•
9th Grade

119 questions
Ch. 6 L. 1 Atoms, Elements & Compounds
Lesson
•
7th - 12th Grade

111 questions
Klasifikasi 5 Kingdom
Lesson
•
10th Grade

110 questions
Marine Science Ch 6
Lesson
•
9th Grade

110 questions
Fingerprints Notes
Lesson
•
11th Grade
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Science

10 questions
Exploring the Evolution of the Peppered Moth
Interactive video
•
6th - 10th Grade

35 questions
DNA Structure and Replication
Quiz
•
10th Grade

10 questions
Exploring Trophic Levels and Food Pyramids
Interactive video
•
6th - 10th Grade

10 questions
Symbiotic Relationships
Lesson
•
9th - 12th Grade

17 questions
Sex-Linked, Co and Incomplete Dominance
Quiz
•
9th - 12th Grade

20 questions
Biology - Intro to Genetics
Quiz
•
10th Grade

10 questions
Exploring Earth's Spheres and Their Interactions
Interactive video
•
6th - 10th Grade

25 questions
Types of Chemical Reactions and Balancing Equations
Quiz
•
10th Grade