

Isotopes in Use
Presentation
•
Science
•
11th - 12th Grade
•
Easy
Standards-aligned
Kristy Hilt
Used 9+ times
FREE Resource
7 Slides • 11 Questions
1

Now that you've found the mass of isotopes on your own, let's see how scientists actually look at isotopes!
Isotopes
2
Isotopes: How are they Measured?
A mass spectrometer is an instrument used to separate an element’s isotopes and to measure the relative isotope abundances.
3
Isotopes: How are they Measured?
How does a spectrometer work?
Within the spectrometer, beams made of charged atoms (ions) from one element are passed through a strong magnetic field. The ions are selected to have identical charges and velocities.
As the ions pass through the magnetic field, the ion paths are influenced by the magnetic force.
Ions of greater mass possess greater inertia or a greater tendency to continue in a straight line. Ions of lesser mass are more influenced by the field and demonstrate greater deviation.
VIDEO TIME!: How does a spectrophotometer work?
4
Isotopes: How are they Measured?
What does a spectrometer produce?

Here is an example of a mass spectrometer reading. It displays the result of passing ions of an element through a spectrometer.
*Note the aces
**The Y-axis should say relative percent abundance
5
Isotopes: How are they Measured?
What does this tell us?

This element has 1 isotope with a mass of 27.
Most elements have more than 1 isotope, resulting in more bars on this type of graph. An example is next!
You will need to determine the element then write each isotope in isotope notation.
6
Isotopes: How are they Measured?
Now you try.
Check it out. Questions coming on the next two slides.
Identify the element.
Isotope Notation.

7
Fill in the Blank

Name the element.
8
Draw
Represent each isotope in proper isotope notation.
9
Isotopes: How are they Measured?
Another Example.
Check it out. Questions coming on the next two slides.
Identify the element.
Isotope Notation.

10
Fill in the Blank

Name the element.
11
Draw
Represent each isotope in proper isotope notation.
12
Multiple Select
Select all that is true.
Isotopes of the same atom have...
the same number of protons.
the same mass.
different quantities of neutrons.
different masses.
13
Multiple Choice
How many protons does P-30 have?
14
Multiple Choice
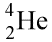
15
Multiple Choice
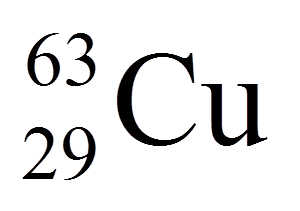
16
Multiple Choice
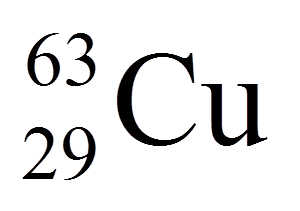
17
Multiple Choice
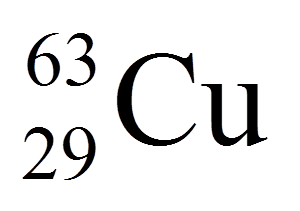
18
Multiple Choice


Now that you've found the mass of isotopes on your own, let's see how scientists actually look at isotopes!
Isotopes
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

15 questions
AP Bio DNA & RNA Structure, & DNA Replication Review
Lesson
•
11th - 12th Grade

14 questions
Earth System and subsystems
Lesson
•
11th - 12th Grade

12 questions
All About Animals
Lesson
•
KG

17 questions
Genetics Review Part 2 (Practice)
Lesson
•
6th - 9th Grade

16 questions
Intro to A&P
Lesson
•
11th - 12th Grade

12 questions
Anne Frank Character Change
Lesson
•
6th - 9th Grade

16 questions
Fats and Oils
Lesson
•
11th - 12th Grade

14 questions
Momentum Practice
Lesson
•
11th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

13 questions
SMS Cafeteria Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

12 questions
SMS Restroom Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

10 questions
Pi Day Trivia!
Quiz
•
6th - 9th Grade
Discover more resources for Science

33 questions
Unit 5 Chemical Reactions Review
Quiz
•
9th - 12th Grade

24 questions
U8 Respiratory System Review
Quiz
•
12th Grade

9 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

17 questions
Sex-Linked, Co and Incomplete Dominance
Quiz
•
9th - 12th Grade

6 questions
Adulting 101: Financial Reality Quiz
Quiz
•
9th - 12th Grade

19 questions
U7 Test: Matter and its Properties
Quiz
•
9th - 12th Grade

13 questions
Understand Mitosis Stages and Functions
Quiz
•
9th - 12th Grade

9 questions
The Mole Concept
Lesson
•
9th - 12th Grade