

Gas Laws - Dalton's Partial Pressure
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Michaela Bratsch
Used 92+ times
FREE Resource
7 Slides • 10 Questions
1
Gas Laws - Dalton's Law of Partial Pressure

2

3

4
5
6

7

8
Multiple Choice
Dalton's law equation is used to find what?
the density of a pure gas
the total pressure of a pure gas
the total pressure of a gas mixture
the volume of a gas mixture
9
Multiple Choice
What does gas pressure depend on?
size of the container and type of gas
number of particles and average kinetic energy
size of container and average mass
the type of gas in the container
10
Multiple Choice
What variables must be kept constant in Dalton's law?
the number of moles
liters and volume
temperature and volume
temperature only
11
Multiple Choice
In a mixture of gases, each gas contributes to the total pressure. The individual contribution of each gas is known as the _________________.
partial volume
partial pressure
molar mass
mole ratio
12
Multiple Choice
A tank of gas contains 50 kPa of helium, 50 kPa of argon, and 100 kPa of neon. What is the total pressure of the gas mixture?
100 kPa
50 kPa
200 L
200 kPa
13
Multiple Choice
A container holds a mixture of two different gases. The oxygen in a container exerts 80 mmHg of pressure on the inside of the container. The total pressure inside the container is 120 mmHg. What is the pressure of the other gas in the container?
200 mmHg
80 mmHg
40 mmHg
120 mmHg
14
Multiple Choice
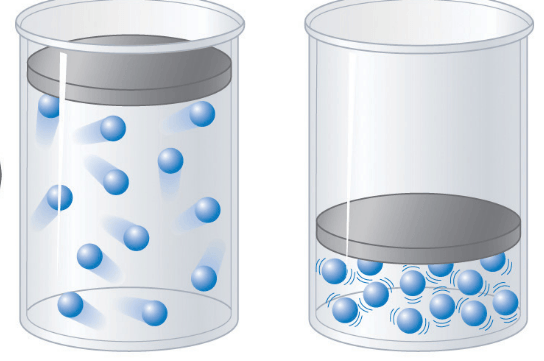
Which container will have a lower pressure?
left
right
they both have the same pressure
I don't know
15
Multiple Choice
A mixture of hydrogen, nitrogen, and water vapor has a total pressure of 864 mmHg. The partial pressure of hydrogen is 220 mmHg and that of nitrogen is 410 mmHg. What is the partial pressure of water vapor?
234 mmHg
1.37 mmHg
2.64 mmHg
1, 494 mmHg
16
Multiple Choice
Three gases, Ar, N2 and H2 are mixed in a 500L container. Ar has a pressure of 255 torr, N2 has a pressure of 228torr and H2 has pressure of 752torr. What is the total pressure in the container?
483torr
270torr
1235torr
17
Multiple Choice
Dalton's Law states...
that at a constant temperature the volume of a confined ideal gas varies inversely with its pressure.
that the total pressure exerted by the mixture of non-reactive gases is equal to the sum of the partial pressures of individual gases
how gases tend to expand when heated
that the density of an ideal gas at constant pressure varies inversely with the absolute temperature of the gas.
Gas Laws - Dalton's Law of Partial Pressure

Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

14 questions
Periodic Table and Trends
Lesson
•
10th - 12th Grade

12 questions
Entropy
Lesson
•
9th - 12th Grade

12 questions
Experience 3: Atomic Emission Spectra
Lesson
•
9th - 12th Grade

13 questions
Classification of Matter
Lesson
•
9th - 12th Grade

11 questions
Intermolecular Forces
Lesson
•
9th - 12th Grade

15 questions
Nuclear Fission vs Nuclear Fusion
Lesson
•
9th - 12th Grade

15 questions
Reading solubility curves
Lesson
•
9th - 12th Grade

14 questions
Particle Diagrams
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

13 questions
SMS Cafeteria Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

12 questions
SMS Restroom Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

10 questions
Pi Day Trivia!
Quiz
•
6th - 9th Grade
Discover more resources for Chemistry

13 questions
Solubility Curves
Quiz
•
10th Grade

10 questions
Exploring Nuclear Fission and Fusion
Interactive video
•
6th - 10th Grade

5 questions
Exit Check 4.1 - Balancing Chemical Equations
Quiz
•
10th Grade

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

8 questions
Labeling a Reaction
Quiz
•
9th - 12th Grade

92 questions
Chem Quarter 3 Review
Quiz
•
10th Grade

19 questions
Isotopes & Ions
Quiz
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade