
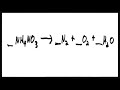
Balancing Chemical Equations Quiz
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard

Nancy Jackson
FREE Resource
5 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in balancing a chemical equation?
Add random coefficients to balance the equation.
Balance oxygen first.
Start with the element that appears most frequently.
Assign coefficients of zero to all species.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why should you avoid starting with oxygen when balancing this equation?
Oxygen is not present in the reactants.
Oxygen appears twice on the product side.
Oxygen is a noble gas.
Oxygen is already balanced.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you determine the coefficient for hydrogen in the equation?
By ensuring the number of hydrogen atoms is equal on both sides.
By doubling the number of hydrogen atoms on the reactant side.
By halving the number of hydrogen atoms on the product side.
By adding one more hydrogen atom to the reactant side.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of multiplying all coefficients by the denominator?
To decrease the number of products.
To increase the number of reactants.
To ensure the equation is balanced chemically.
To simplify the equation.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final step in balancing the chemical equation?
Add more reactants to the equation.
Ensure the equation is balanced chemically by adjusting coefficients.
Check if all elements are balanced mathematically.
Remove excess products from the equation.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
