
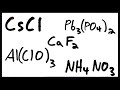
Ionic Compounds Formula Writing Quiz
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard

Nancy Jackson
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in writing the formula for an ionic compound?
Find the least common multiple of the charges
Adjust the subscripts to balance charges
Write the formula for the anion first
Write the formulas for the cation and anion separately
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the cation written first in the formula of an ionic compound?
It is heavier than the anion
It is more reactive than the anion
It's a convention to write the positively charged ion first
Because it has a higher atomic number
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the cesium ion in cesium chloride?
+1
+2
-1
-2
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the formula for calcium fluoride, why is the subscript for fluoride doubled?
To match the atomic mass of calcium
To balance the charges between calcium and fluoride
To make the formula more stable
To increase the solubility of the compound
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the calcium ion in calcium fluoride?
+1
+2
-1
-2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula for the ammonium ion?
NH2-
NO3-
NH4+
NH3
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a polyatomic ion?
NH4+
O2-
Cl-
Na+
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
