
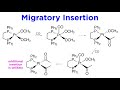
Platinum Complex Reactions and Mechanisms
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a necessary condition for migratory insertion to occur between two ligands?
The ligands must be identical.
The ligands must be cis to each other.
The ligands must be trans to each other.
The ligands must be in different coordination spheres.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which type of bond is most commonly involved in migratory insertion?
Sigma bond
Delta bond
Pi bond
Ionic bond
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the context of migratory insertion, what happens when a strong ligand binds to the vacant site?
The ligands become trans to each other.
The insertion becomes irreversible.
The insertion becomes reversible.
The oxidation state of the metal changes.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of a hydrometalation reaction?
An alkene becomes an alkyl ligand.
A metal-carbon bond becomes a pi bond.
A ligand dissociates from the metal.
A metal-oxygen bond is formed.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary outcome of carbometalation?
Formation of a nitrile ligand
Formation of a hydride ligand
Formation of a propyl ligand
Formation of a benzyl ligand
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a stable iron complex with 18 electrons, what is unlikely to occur?
Coordination of triphenylphosphine
Ligand dissociation
Change in oxidation state
Migratory insertion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the preferred electron count for a stable square planar platinum complex?
20 electrons
16 electrons
18 electrons
14 electrons
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
