
Valence Electrons and Lewis Structures
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
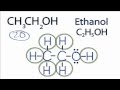
What is another name for the compound CH3CH2OH?
Methanol
Ethanol
Butanol
Propanol
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many total valence electrons are present in the Lewis structure of ethanol?
22
18
24
20
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of drawing lines between atoms in a Lewis structure?
To represent atomic mass
To show chemical bonds
To display molecular weight
To indicate electron spin
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons are used to form chemical bonds in the initial skeleton structure of ethanol?
18
16
14
12
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the octet rule in the Lewis structure of ethanol?
It calculates the molecular weight
It ensures atoms have full outer shells
It predicts the boiling point
It determines the molecular shape
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons does each hydrogen atom have in the ethanol Lewis structure?
Three
Four
Two
One
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
