
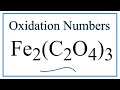
Oxidation Numbers and Charges
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the overall charge of the compound Iron(III) Oxalate?
Positive
Variable
Negative
Neutral
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the oxalate ion (C2O4)?
1+
3+
0
2-
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the oxidation numbers in an ion relate to the ion's charge?
They are always negative.
They add up to the ion's charge.
They add up to zero.
They are always positive.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If there are three oxalate ions, what is the total oxidation number for these ions?
-3
0
-6
+6
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What oxidation number must each iron atom have to balance the compound?
+4
+6
+3
+2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the sign of an oxidation number written compared to an ionic charge?
After the number for both.
After the number for oxidation numbers, before for ionic charges.
Before the number for both.
Before the number for oxidation numbers, after for ionic charges.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the oxidation number of iron in Iron(III) Oxalate?
+2
+3
+4
+5
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
