
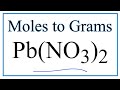
Moles and Grams Conversion Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial quantity of lead(II) nitrate given in moles?
0.2 moles
0.4 moles
0.6 moles
0.8 moles
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in converting moles to grams?
Subtract the molar mass
Add the molar mass
Multiply by the molar mass
Divide by the molar mass
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of lead(II) nitrate used in the calculation?
300.5 grams per mole
250.3 grams per mole
331.2 grams per mole
400.1 grams per mole
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
After multiplying the moles by the molar mass, what is the resulting unit?
Kilograms
Grams
Liters
Moles
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final mass of lead(II) nitrate obtained from the conversion?
100.5 grams
132.5 grams
200.0 grams
150.0 grams
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If you start with grams and want to find moles, what operation should you perform?
Multiply by the molar mass
Divide by the molar mass
Subtract the molar mass
Add the molar mass
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Who is the presenter of the video?
Dr. D
Dr. C
Dr. B
Dr. A
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
