
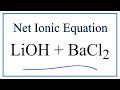
Chemical Reactions and Ionic Equations
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in writing a net ionic equation?
Determine the solubility of compounds
Balance the molecular equation
Write the ionic equation
Identify spectator ions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you balance the chlorine atoms in the equation?
Add a coefficient of 2 in front of Ba(OH)2
Add a coefficient of 2 in front of LiCl
Add a coefficient of 2 in front of BaCl2
Add a coefficient of 2 in front of LiOH
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the state of lithium hydroxide in the reaction?
Aqueous
Gas
Liquid
Solid
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to check the solubility table for barium hydroxide?
To determine if it is a gas
To confirm if it is a strong acid
To check if it reacts with lithium
To verify if it is soluble or forms a solid
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the ions in the reaction?
They form a precipitate
They dissolve completely
They remain unchanged
They react to form a gas
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of crossing out the spectator ions?
A net ionic equation is obtained
A new compound is formed
A precipitate is formed
No ions are left, indicating no reaction
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is there no net ionic equation for the reaction?
All substances are gases
The reaction produces a new compound
All ions are unchanged and remain aqueous
A solid precipitate forms
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
