
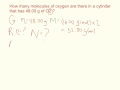
Calculating Moles and Molecules
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial problem discussed in the video?
Measuring the pressure of a gas
Determining the number of oxygen molecules in a cylinder
Calculating the volume of a gas
Finding the number of oxygen atoms in a cylinder
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the molar mass of O2 calculated?
By multiplying the atomic mass of oxygen by 2
By dividing the atomic mass of oxygen by 2
By subtracting the atomic mass of hydrogen from oxygen
By adding the atomic numbers of two oxygen atoms
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass of a single oxygen atom used in the calculation?
16.00
8.00
12.00
32.00
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the number 2 in the O2 molecule?
It shows the number of protons
It indicates the number of oxygen atoms in the molecule
It represents the number of electrons
It is the charge of the molecule
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the term 'entities' refer to in the context of this video?
Ions
Molecules
Atoms
Compounds
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula used to calculate moles from mass and molar mass?
Moles = Mass x Molar Mass
Moles = Molar Mass / Mass
Moles = Mass + Molar Mass
Moles = Mass / Molar Mass
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the calculated number of moles for 48.00 grams of O2?
1.0 moles
2.5 moles
1.5 moles
2.0 moles
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
