
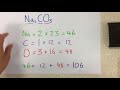
Relative Formula Mass Calculations
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary goal of finding the relative formula mass?
To determine the weight of a single atom
To find the relative mass of a molecule
To measure the volume of a liquid
To calculate the density of a substance
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example of sodium carbonate (Na2CO3), how many sodium atoms are present?
Three
Two
Four
One
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass of carbon as used in the sodium carbonate example?
40
23
12
16
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the mass of oxygen used in the sodium carbonate calculation?
40
23
16
12
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you determine the number of atoms in a molecule when there are brackets involved?
Multiply the number of atoms inside the brackets by the number outside
Add the numbers inside and outside the brackets
Count each atom individually
Ignore the brackets
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relative formula mass of calcium hydroxide (Ca(OH)2)?
32
40
106
74
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it recommended to use a calculator even for simple additions?
To ensure accuracy
To practice calculator skills
To save time
To avoid mental math
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
