
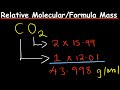
Relative Atomic and Molecular Mass Concepts
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relative atomic mass of an element based on?
The chemical reactivity of the element
The number of protons in the nucleus
The weighted average of isotopes compared to carbon-12
The mass of a single atom
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it difficult to measure the mass of a single atom directly?
Atoms are constantly moving
Atoms have no mass
Atoms are too large to fit on a scale
Atoms are too small to be weighed directly
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass unit (amu) equivalent to in kilograms?
1.00 x 10^-3 kg
6.022 x 10^23 kg
1.66 x 10^-24 kg
1.66 x 10^-27 kg
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the relative molecular mass of a compound determined?
By multiplying the atomic masses of all atoms
By dividing the total mass by the number of atoms
By adding the atomic numbers of all atoms
By summing the relative atomic masses of all atoms in the molecule
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relative molecular mass of carbon dioxide (CO2)?
15.994
44.01
43.998
12.01
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relative formula mass of a compound?
The sum of atomic numbers in the compound
The sum of relative atomic masses in the compound
The product of atomic masses in the compound
The average mass of the compound's isotopes
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the formula for calcium hydroxide, what does the subscript '2' indicate?
Two oxygen atoms
Two hydroxide ions
Two hydrogen atoms
Two calcium atoms
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
