
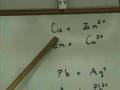
Reactivity and Spontaneity in Chemistry
Interactive Video
•
Chemistry, Science, Biology
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of Table J in chemistry?
To list elements alphabetically
To show the reactivity order of elements
To display atomic weights
To categorize elements by color
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why are group one metals considered highly reactive?
They have high electronegativities
They are located at the bottom of the periodic table
They have multiple valence electrons
They easily donate their single valence electron
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which element is known for having the highest desire for electrons?
Chlorine
Oxygen
Fluorine
Nitrogen
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a spontaneous reaction mean in practical terms?
It only occurs at high temperatures
It occurs without additional energy once started
It requires continuous energy input
It never occurs naturally
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a non-spontaneous process?
A candle burning
A battery being recharged
Ice melting at room temperature
A battery powering a device
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a spontaneous reaction, which element should be in the elemental state?
The element with the highest atomic number
The element higher in the chart
The element lower in the chart
The element with the lowest atomic weight
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does zinc react spontaneously with copper ions?
Zinc is less reactive than copper
Copper has more electrons to donate
Copper is higher in the reactivity series
Zinc is higher in the reactivity series
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
