
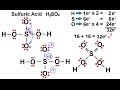
Understanding H2SO4 Structure and Bonding
Interactive Video
•
Chemistry, Science, Biology
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary reason for the octet rule violation in the H2SO4 molecule?
Lack of valence electrons
High electronegativity of sulfur
Separation of formal charges
Presence of hydrogen atoms
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons are available for bonding in the H2SO4 molecule?
24
28
32
36
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atom is identified as the central atom in the H2SO4 molecule?
Oxygen
Hydrogen
None of the above
Sulfur
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What formal charge does sulfur initially have in the H2SO4 molecule?
0
+1
+2
-2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the initial bonding scenario in H2SO4 considered unstable?
Too many electrons
Separation of formal charges
Lack of hydrogen bonds
Excessive double bonds
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What structural change is made to stabilize the H2SO4 molecule?
Increasing the number of valence electrons
Removing oxygen atoms
Forming double bonds with sulfur
Adding more hydrogen atoms
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many double bonds are formed in the stable structure of H2SO4?
One
Two
Three
Four
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
