
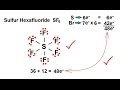
Sulfur Hexafluoride Bonding Concepts
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main focus of the video tutorial?
The octet rule in chemistry
Molecules that do not follow the octet rule
The structure of water molecules
The periodic table of elements
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons does sulfur have?
Seven
Four
Six
Eight
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is fluorine eager to form a single bond?
To gain an electron
To share electrons
To lose electrons
To gain stability
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of sulfur in the sulfur hexafluoride molecule?
It remains unbonded
It acts as a central atom forming six bonds
It donates electrons to fluorine
It forms double bonds with fluorine
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many bonds does sulfur form in sulfur hexafluoride?
Four
Seven
Five
Six
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the electron arrangement in each fluorine atom in sulfur hexafluoride?
A sextet
A doublet
A triplet
An octet
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the term used to describe sulfur's electron arrangement in sulfur hexafluoride?
Standard octet
Expanded octet
Incomplete octet
Double octet
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
