
Atomic Mass and Isotopes Concepts
Interactive Video
•
Chemistry, Physics, Mathematics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the standard element used to define atomic mass?
Nitrogen-14
Carbon-12
Oxygen-16
Hydrogen-1
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is AMU preferred over other units for atomic mass?
It is the only unit available.
It is easier to use and understand.
It is more accurate.
It is used only for carbon.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What distinguishes isotopes of the same element?
Different numbers of electrons
Different numbers of protons
Different numbers of neutrons
Different atomic numbers
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the average atomic mass on the periodic table determined?
By using a weighted average
By using the most common isotope
By using a simple average
By listing all isotopes
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a real-world example used to explain weighted averages?
Counting stars
Measuring temperature
GPA in school
Calculating speed
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
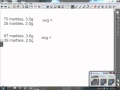
In the marble example, why is the average not simply the mean of the two weights?
Because the weights are not in grams
Because the marbles are not round
Because the marbles are different colors
Because the weights are not equal
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the key factor in calculating a weighted average for isotopes?
The abundance of each isotope
The color of the isotope
The charge of the isotope
The size of the isotope
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
