
Mole Conversions in Chemistry
Interactive Video
•
Chemistry, Science, Mathematics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary role of the mole in chemistry?
A unit of time
A unit of pressure
A unit of amount and conversion
A unit of temperature
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
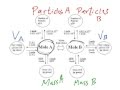
Which of the following is NOT a direct conversion using the mole conversion diagram?
Volume to mass
Mass to particles
Volume to volume
Volume to particles
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the mole conversion diagram, what must you always pass through to convert between different units?
Mass
Particles
Moles
Volume
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the importance of following the arrows in the mole conversion diagram?
To memorize the periodic table
To avoid using a calculator
To make the diagram look neat
To ensure correct conversion
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the symbol for elemental gold used in the example?
Fe
Au
Ag
O
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many grams of gold are used in the example problem?
20 grams
15 grams
10 grams
25 grams
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the atomic mass of gold as used in the calculation?
196.9
197.0
198.0
199.0
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
