
Electrolytes and Ionization Concepts
Interactive Video
•
Chemistry, Science, Biology
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary role of free ions in an electrolyte?
To form solid compounds
To conduct electricity
To evaporate quickly
To dissolve in water
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to glucose when it is dissolved in water?
It forms ions
It remains as glucose surrounded by water molecules
It evaporates
It turns into a gas
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of electrolyte is formed when sodium chloride is dissolved in water?
Non-electrolyte
Weak electrolyte
No electrolyte
Strong electrolyte
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a single reaction arrow indicate in a chemical equation?
Reversible reaction
Partial conversion of reactants to products
Complete conversion of reactants to products
No reaction occurs
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
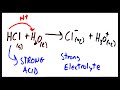
What happens when HCl is dissolved in water?
It forms a strong electrolyte
It forms a weak electrolyte
It does not dissolve
It forms a non-electrolyte
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of molecular ionization with a strong acid?
Formation of a non-electrolyte
Formation of a strong electrolyte
No ion formation
Formation of a weak electrolyte
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What distinguishes a weak acid from a strong acid in terms of ion formation?
Weak acids form ions rapidly
Weak acids form ions partially
Weak acids do not form ions
Weak acids form ions completely
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
