
Ionic Bonds and Noble Gas Stability Explained
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the electronic configuration of neon?
2, 8
2, 6
2, 8, 8
2, 2
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why are noble gases considered stable?
They can easily gain electrons.
They have a single electron in their outer shell.
They have no electrons.
They have a full outer shell of electrons.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the octet rule?
Atoms are stable with no electrons in their outer shell.
Atoms are stable with ten electrons in their outer shell.
Atoms are stable with eight electrons in their outer shell.
Atoms are stable with two electrons in their outer shell.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
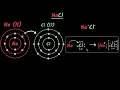
How does sodium achieve stability?
By losing one electron.
By gaining one electron.
By gaining seven electrons.
By losing seven electrons.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to chlorine when it gains an electron?
It loses its charge.
It remains neutral.
It becomes positively charged.
It becomes negatively charged.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of sodium transferring an electron to chlorine?
No bond is formed.
Formation of a metallic bond.
Formation of a covalent bond.
Formation of an ionic bond.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the electron dot structure used in representing ionic bonds?
By showing the protons in the nucleus.
By showing the nucleus of atoms.
By showing the transfer of valence electrons.
By showing the sharing of electrons.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
