
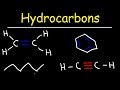
Hydrocarbon Structures and Formulas
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Easy
Sophia Harris
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a characteristic feature of aromatic hydrocarbons?
They contain only single bonds.
They have a benzene ring structure.
They are saturated with hydrogen.
They are always linear.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a saturated hydrocarbon?
Ethene
Ethyne
Ethane
Benzene
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the general formula for alkanes?
CnH2n+1
CnH2n+2
CnH2n
CnH2n-2
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which alkane has the molecular formula C5H12?
Butane
Heptane
Pentane
Hexane
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the general formula for alkenes with one double bond?
CnH2n
CnH2n+2
CnH2n-2
CnH2n+1
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the presence of a double bond affect the hydrogen count in alkenes?
It increases the hydrogen count by two.
It decreases the hydrogen count by two.
It has no effect on the hydrogen count.
It doubles the hydrogen count.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the general formula for alkynes with one triple bond?
CnH2n
CnH2n+2
CnH2n-2
CnH2n+1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
