
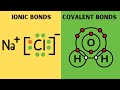
Understanding Ionic and Covalent Bonds
Interactive Video
•
Chemistry, Science
•
6th - 9th Grade
•
Practice Problem
•
Medium
Liam Anderson
Used 145+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary difference between ionic and covalent bonds?
Ionic bonds involve sharing electrons, while covalent bonds involve transferring electrons.
Ionic bonds involve transferring electrons, while covalent bonds involve sharing electrons.
Both ionic and covalent bonds involve sharing electrons.
Both ionic and covalent bonds involve transferring electrons.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a characteristic of ionic bonds?
Formed between two non-metals
Formed between metals and non-metals
Involves sharing of electrons
Involves no electron movement
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How can you identify a covalent bond using the periodic table?
Look for elements to the right of the stair step
Look for a combination of metals and non-metals
Look for elements to the left of the stair step
Look for a combination of non-metals
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following compounds is an example of an ionic bond?
Sodium chloride
Water
Hydrogen gas
Sulfur dioxide
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a polyatomic ion?
A group of non-metals bonded together acting as one
A group of metals bonded together
A single element with a charge
A metal bonded with a non-metal
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a covalent bond?
Potassium bromide
Lithium nitrate
Sulfur dioxide
Sodium chloride
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How are atoms held together in covalent bonds?
By transferring electrons
By sharing electrons
By magnetic forces
By gravitational forces
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
