
Exploring Lewis Acids and Bases in Organic Chemistry
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Hard
Liam Anderson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary characteristic of a Lewis acid?
Electron pair donor
Electron pair acceptor
Proton donor
Proton acceptor
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a Lewis base?
H+
Fe2+
NH3
Al3+
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does it mean if a substance is amphoteric?
It can only donate electrons
It can only accept electrons
It can act as both an acid and a base
It does not participate in reactions
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which molecule is described as having a trigonal planar shape in its reaction?
BF3
FeCl3
AlBr3
ZnCl2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
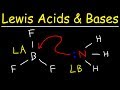
In the reaction between BF3 and NH3, what is the charge on boron in the product?
Positive
Negative
Neutral
Variable
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the product when FeCl3 reacts with Cl-?
FeCl2
FeCl4-
Fe2Cl3
FeCl3+
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the formal charge of zinc in the reaction with water?
Becomes positive
Becomes negative
Remains neutral
Becomes variable
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
