
Exploring Isotopes and Ions
Interactive Video
•
Chemistry
•
6th - 10th Grade
•
Practice Problem
•
Medium
Emma Peterson
Used 1+ times
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the standard form of an element on the periodic table represent?
An element with a different number of electrons
The most common form of the element
An element with a different number of protons
An element with a different number of neutrons
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What changes in an isotope of an element?
Atomic number
Number of neutrons
Number of electrons
Number of protons
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is an isotope of an element written differently from its standard form?
Elemental symbol followed by a dash and the new atomic number
Elemental symbol followed by a dash and the new atomic mass number
Elemental symbol followed by a dash and the new number of protons
Elemental symbol followed by a dash and the new number of electrons
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can isotopes be dangerous?
They emit subatomic particles
They change the atomic number
They have fewer electrons than usual
They have more protons than usual
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
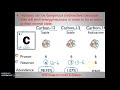
What is the most common isotope of carbon found on Earth?
Carbon-12
Carbon-13
Carbon-14
Carbon-15
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is an ion?
An atom that has acquired a full charge
An atom with a different number of neutrons
An atom with a different atomic number
An atom with a different number of protons
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a cation?
An ion with a negative charge
An ion with an extra neutron
An ion with a positive charge
An ion with no charge
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
