
Quantum Mechanical Model of the Atom
Authored by Lisa Thompson
Science
9th Grade
NGSS covered
Used 3+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
25 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
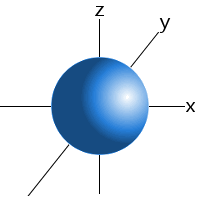
Which shape represents the shape of an "s" orbital?
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
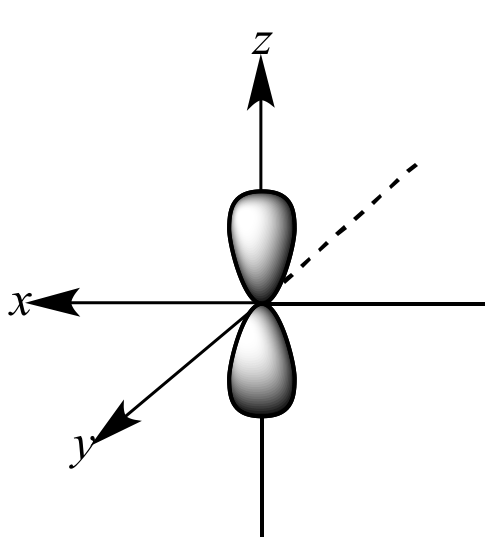
Which shape represents the shape of an "p" orbital?



3.
OPEN ENDED QUESTION
1 min • 1 pt

Compare and contrast the Bohr Model and the Quantum Model of the atom. Describe at least two key similarities and at least one key difference.
Evaluate responses using AI:
OFF
4.
MULTIPLE SELECT QUESTION
1 min • 1 pt
There are four different orbital shapes. Select them from the following list.
"s"
"f"
"g"
"d"
"p"
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
In the quantum mechanical model of the atom, where are electrons most likely to be found?
Moving in fixed orbits around the nucleus
Embedded within the nucleus
In specific regions called electron clouds or orbitals
Uniformly distributed around the nucleus
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
In the quantum-mechanical model of the atom, an orbital is defined as a
region of the most probable proton location.
region of the most probable electron location.
circular path traveled by an electron around an orbital.
circular path traveled by a proton around an orbital.
Answer explanation
In the quantum-mechanical model of the atom, an orbital is defined as a region of the most probable electron location.
7.
MULTIPLE SELECT QUESTION
1 min • 1 pt
What is the fundamental difference between the Bohr model of an atom and the quantum mechanical model? Select all that apply.
Bohr model considers electron to be a particle; quantum mechanics considers it to be a wave.
Bohr model describes electron with 1 quantum number; quantum model uses 4 quantum.
Bohr model describes electrons in orbits; quantum model describes electrons in orbitals.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
