
Lewis Dot and Bonding
Authored by Elizabeth Fredrickson
Chemistry
9th - 12th Grade
NGSS covered
Used 36+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
50 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
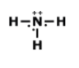
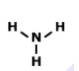
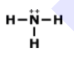
What is the correct Lewis Dot Structure for ammonia NH3
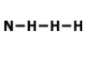
Tags
NGSS.MS-PS1-1
2.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Which is the correct molecular structure for carbon dioxide?




Tags
NGSS.MS-PS1-1
3.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt

How many total valence electrons are participating in bonding in the molecule above?
8
4
2
3
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
4.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt

Which of the following is the correct Lewis dot structure for the molecule fluorine (F2)?
Tags
NGSS.MS-PS1-1
5.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
NH3 has how many lone pairs?
0
1
2
3
Tags
NGSS.MS-PS1-1
6.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
CCl4 has how many double bonds?
0
1
2
3
Tags
NGSS.HS-PS1-1
7.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Carbonate (CO32-) has how many double bonds?
0
1
2
3
Tags
NGSS.HS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

54 questions
Experience Chemistry Investigation 2 Test Review
Quiz
•
10th - 12th Grade

50 questions
Chemistry Fall Review
Quiz
•
10th Grade

46 questions
Speed Dating Elements
Quiz
•
10th Grade

47 questions
Ionic Bonding
Quiz
•
10th - 11th Grade

47 questions
Enlace químico
Quiz
•
10th Grade

50 questions
Rate of reaction/Kadar tindakbalas
Quiz
•
10th Grade

50 questions
Q3 Review 2023
Quiz
•
10th Grade

50 questions
Chemical Bonds-AH
Quiz
•
9th Grade - University
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

19 questions
Stoichiometry, Limiting Reactants, and Percent Yield
Quiz
•
10th Grade

20 questions
Stoichiometry Practice
Quiz
•
12th Grade

10 questions
Formative 3BD: Ionic Bonds
Quiz
•
9th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

10 questions
Identifying types of reactions
Quiz
•
9th - 12th Grade