
Acids and Bases Strengths
Authored by Charles Martinez
Chemistry
11th - 12th Grade
NGSS covered

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
20 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt

Which of the following best approximates the Ka value for this weak acid?
1 x 10–3
1 x 10–4
1 x 10–5
1 x 10–6
2.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Which of the following best represents a solution of H2SO4 in water?
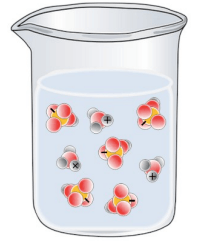
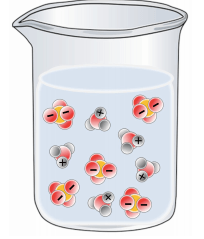

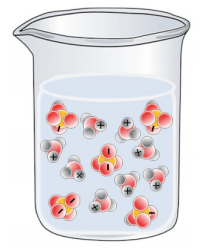
Tags
NGSS.HS-PS1-2
3.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
HSO4¯ + H2O ↔ H3O+ + SO4 2¯ In the equilibrium represented above, the species that act as bases include which of the following?
HSO4-
H2O
SO4 2¯
H2O and SO4 2¯
4.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Given the equation:
H2SO4 + H2O ↔ H3O+ + HSO4- 1
What is the conjugate acid?
H2SO4
H2O
H3O+
HSO4- 1
5.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Which compound is the conjugate base?
HCO3- + HCl ==> H2CO3 + Cl-
Which compound is the conjugate base?
HCO3-
HCl
H2CO3
Cl-
6.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Which of the following is transferred between a conjugate acid-base pair?
an electron
a proton
a neutron
a OH- ion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When a hydrogen ion is lost from an acid, the resulting particle is called its conjugate base. Which of the following particles would be the conjugate base for the acid HSO4–?
H2SO4
SO42–
SO4–
HSO4
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
