
Let's Review: Element Identity & Reactivity
Authored by Victoria Gandy
Chemistry, Science
6th - 8th Grade
NGSS covered
Used 5+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
An atom of which element has 37 protons, 48 neutrons and 1 valence electron?
Neon (Ne)
Carbon (C)
Rubidium (Rb)
Sodium (Na)
Tags
NGSS.HS-PS1-1
2.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
When trying to identify an unknown element, a scientist determines what other elements reacts with chemically. Which property of the unknown element determines the other element it reacts with?
The total number of neutrons in the unknown element
The total number of particles in the nucleus of the unknown element
The number of protons in the nucleus of the unknown element
The number of valence electrons in the unknown element
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
3.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
The atoms of a certain element each contain 9 protons and 7 valence electrons. Which statement correctly identifies this element and describes the chemical reactivity?
The element is oxygen, and is highly reactive.
The element is fluorine, and is highly reactive.
The element is oxygen, and is not very reactive.
The element is fluorine, and is not very reactive.
4.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
Which model represents the most reactive atom?
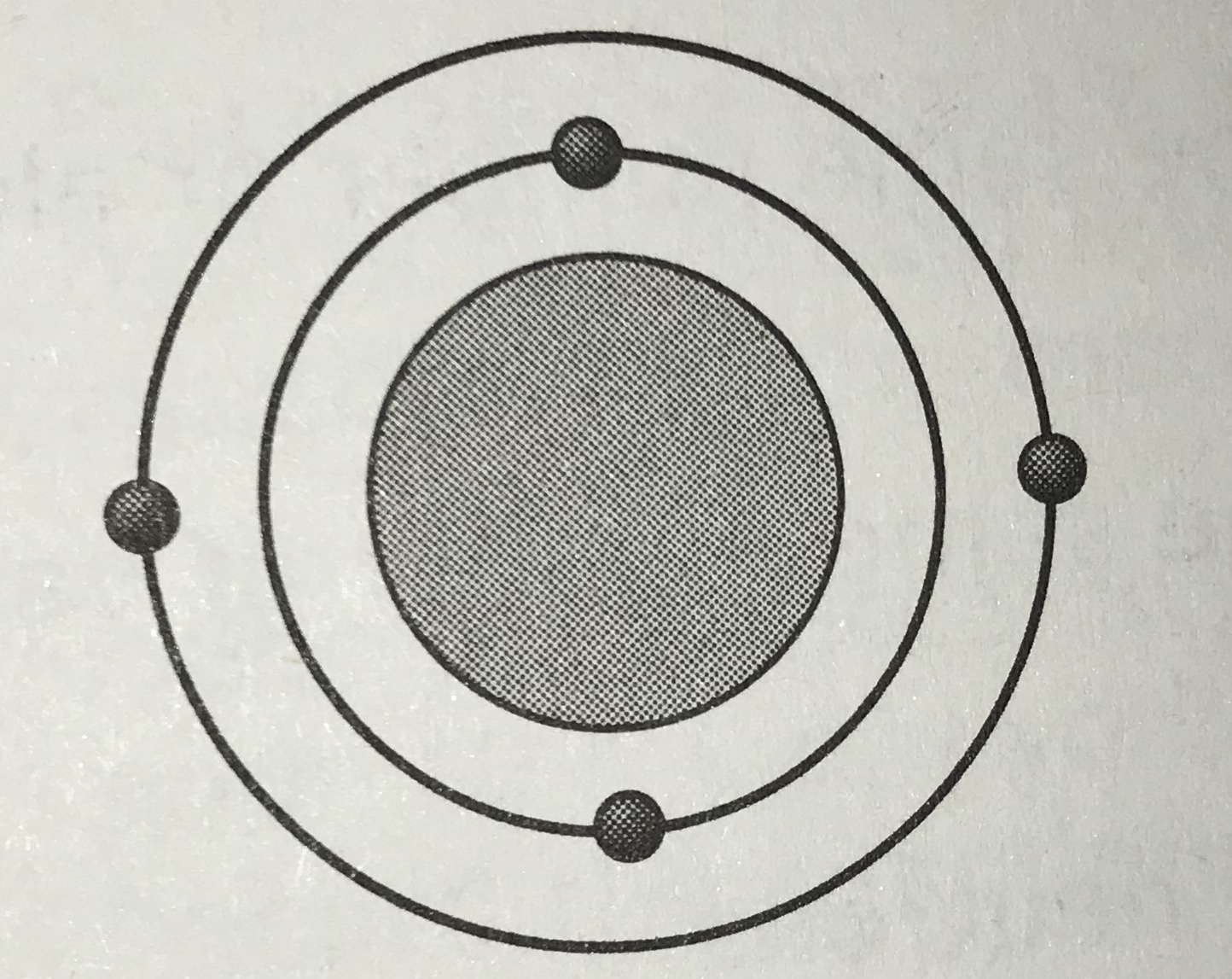
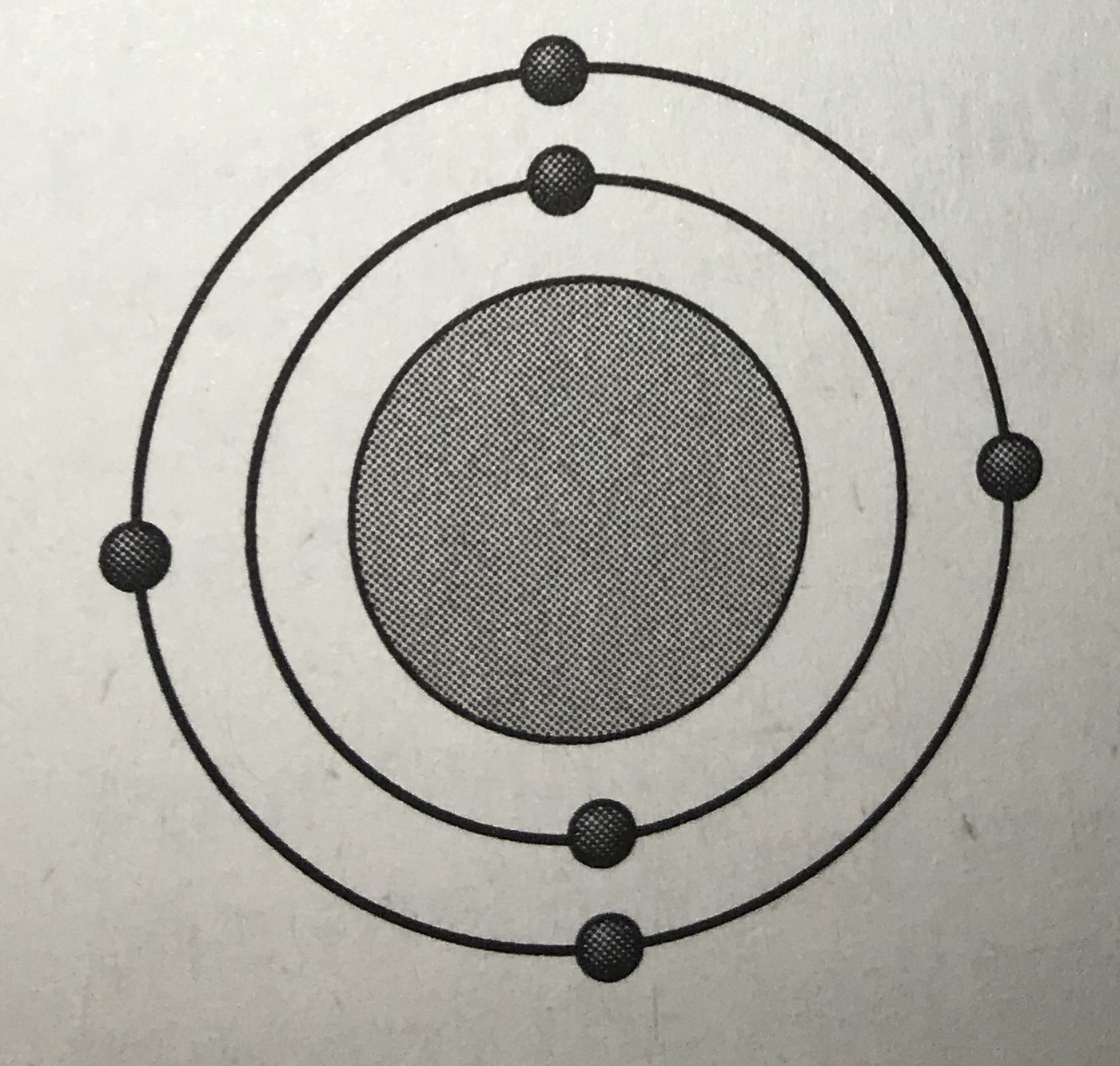
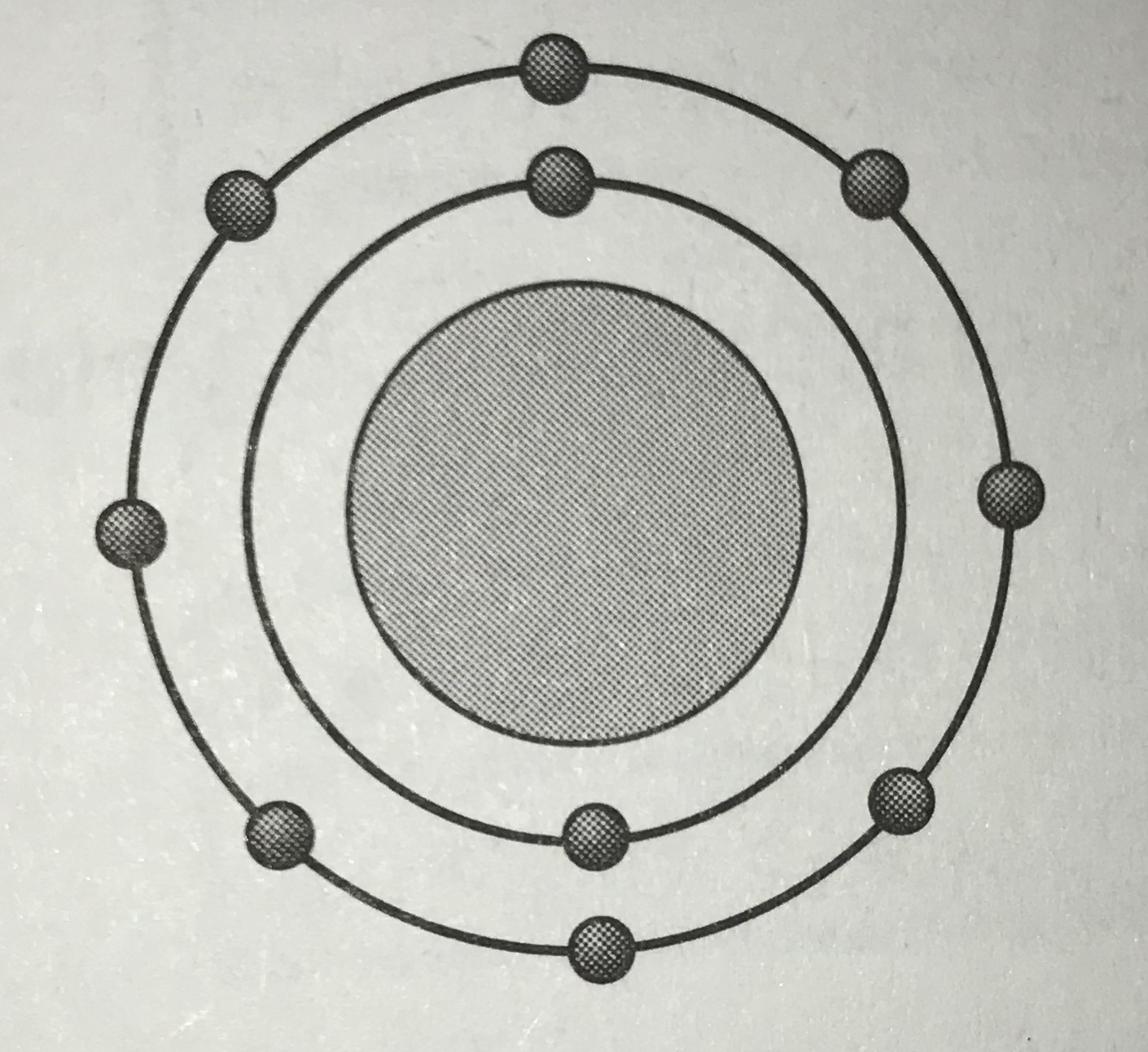
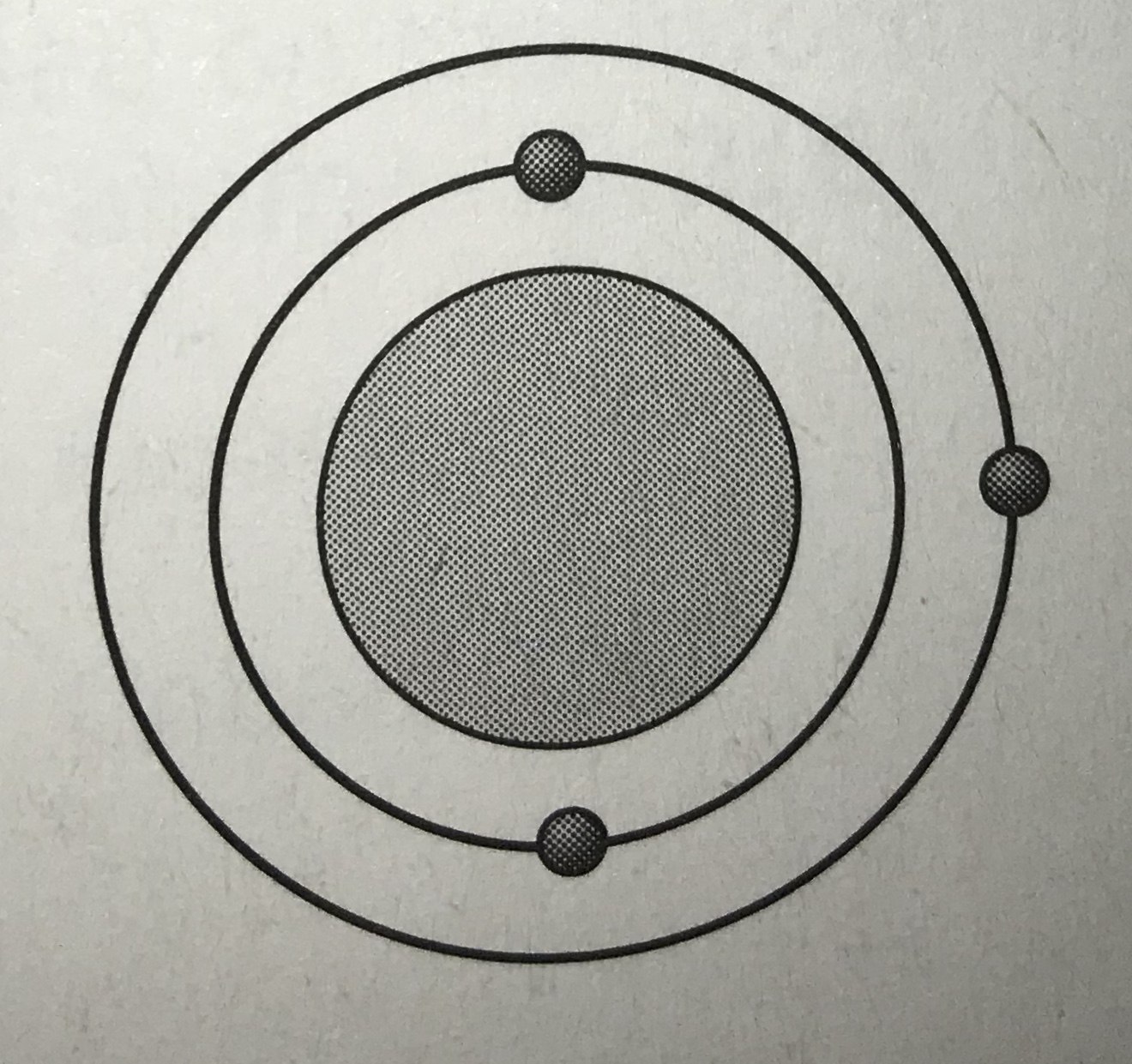
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
5.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
Which of the following characteristics is similar for fluorine and iodine?
Number of protons
Number of neutrons
Number of orbitals
Number of valence electrons
6.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
A group of students examines a Bohr model of an atom with 8 valence electrons. What conclusion can the students make about the chemical reactivity of the element?
The element is extremely reactive because the outer energy level is full
The element is nonreactive because the outer energy level is full
The element is somewhat reactive because the outer energy level is full
Additional information is necessary to determine reactivity
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
7.
MULTIPLE CHOICE QUESTION
5 mins • 1 pt
The atomic number of an element is equal to its number of protons. Therefore, a logical conclusion is that the number of protons determines an element's
Mass and atomic weight
Number of stable isotopes
Identity
State of matter
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?

