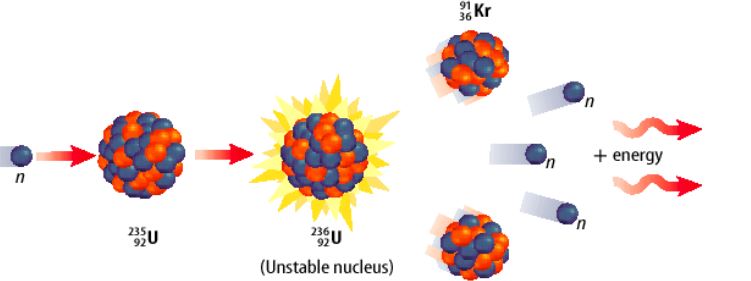
Nuclear Fission & Fusion
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+1
Standards-aligned
Allison Boggess
Used 2+ times
FREE Resource
15 Slides • 29 Questions
1
2
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
10
11
12
13
Multiple Select
14
Multiple Select
15
16
17
18
19
20
Multiple Choice
21
Multiple Choice
22
Multiple Select
23
Multiple Choice
24
25
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Select
30
Multiple Choice
31
Multiple Choice
32
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
43
Categorize
44
Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

36 questions
Timeline of Earth
Lesson
•
9th - 12th Grade

36 questions
Earth Early History
Lesson
•
9th - 12th Grade

35 questions
Evidence of Evolution
Lesson
•
9th - 12th Grade

42 questions
Fossil Fuels
Lesson
•
9th - 12th Grade

43 questions
SOL INTENSIVE REVIEW (BASED ON MOCK SOL TEST-Part 2)
Lesson
•
9th - 12th Grade

38 questions
Intro to Bonds
Lesson
•
9th - 11th Grade

36 questions
7A Review
Lesson
•
9th - 12th Grade

42 questions
7.3 Atmospheric Moisture (Earth Science)
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Science

8 questions
Momentum and Collisions
Lesson
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

10 questions
Exploring Weathering, Erosion, and Deposition Processes
Interactive video
•
6th - 10th Grade

10 questions
Cladogram Practice
Quiz
•
9th Grade

12 questions
The Respiratory System
Lesson
•
9th - 12th Grade

14 questions
Natural Selection and Adaptation
Lesson
•
9th - 12th Grade

10 questions
Exploring the Rock Cycle and Plate Tectonics
Interactive video
•
6th - 10th Grade

15 questions
Chemical Reactions (Types of Chemical Reactions)
Interactive video
•
11th Grade