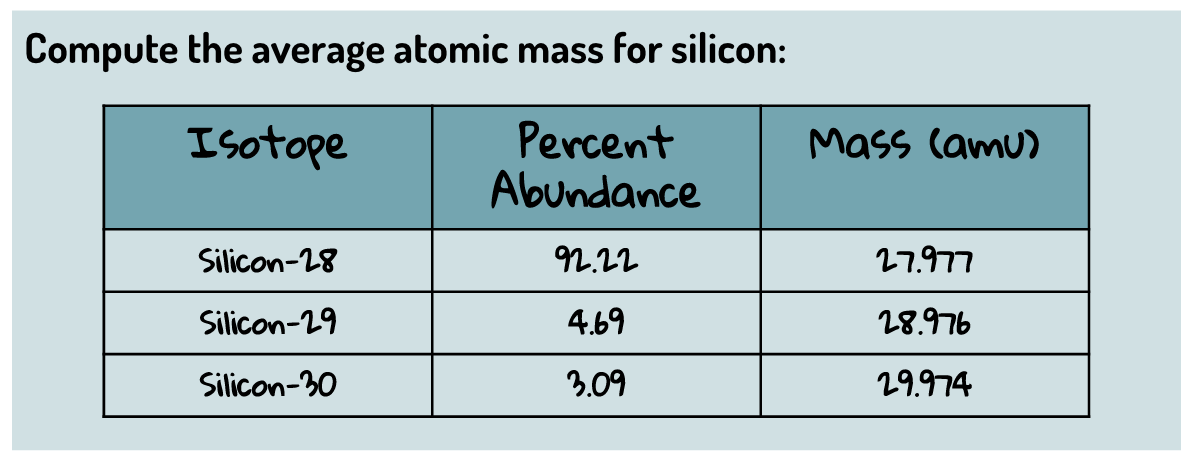
Atomic Structure Day 4 - Calculating Atomic Mass
Presentation
•
Chemistry
•
9th - 12th Grade
•
Easy
Standards-aligned
Kimette Witt
Used 1+ times
FREE Resource
18 Slides • 3 Questions
1
2
3
4
5
6
7
8
9
10
11
Multiple Choice
12
13
14
15
16
17
18
19
Multiple Choice
20
Multiple Choice
21
Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

19 questions
Review Periodic Table
Lesson
•
10th - 12th Grade

21 questions
Isotopes and Avg. Atomic Mass Notes
Lesson
•
9th - 12th Grade

17 questions
3.7 Solutions and Mixtures
Lesson
•
9th - 12th Grade

18 questions
1.3: Elemental Composition of Pure Substances
Lesson
•
9th - 12th Grade

15 questions
TSIA Prep Quantitative Reasoning
Lesson
•
10th - 12th Grade

20 questions
Human Population Growth Lesson
Lesson
•
9th - 12th Grade

15 questions
Energy in Food
Lesson
•
9th Grade - University

17 questions
Experience Chemistry Lesson 12.1-Rates of Reaction
Lesson
•
10th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Energy Review
Quiz
•
9th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade