

kinetic theory of gases
Presentation
•
Science
•
9th Grade
•
Practice Problem
•
Medium
Standards-aligned
April Elmore
Used 5+ times
FREE Resource
0 Slides • 16 Questions
1
Multiple Choice
Which best describes a solid?
The state of matter that has a fixed shape and fixed volume
The molecules of a solid move with a high speed in random order
The state of matter that has a fixed volume and takes the shape of its container
Molecules move past one another, vibrate, and rotate
2
Multiple Choice

Which state of matter is shown?
sold
liquid
gas
plasma
3
Multiple Choice
Which is an assumption of the kinetic theory of gases?
Gas are made up of particles that move at a constant, random motion
Gases can be compressed
Evaporation is a state change
When particles of a gas collide a large amount of energy is lost
4
Multiple Choice
Which is FALSE about melting?
solid change to a liquid
particles gain enough energy to move past each other
the heat of fusion is needed to melt a solid
the heat of vaporization is needed to melt a solid
5
Multiple Choice
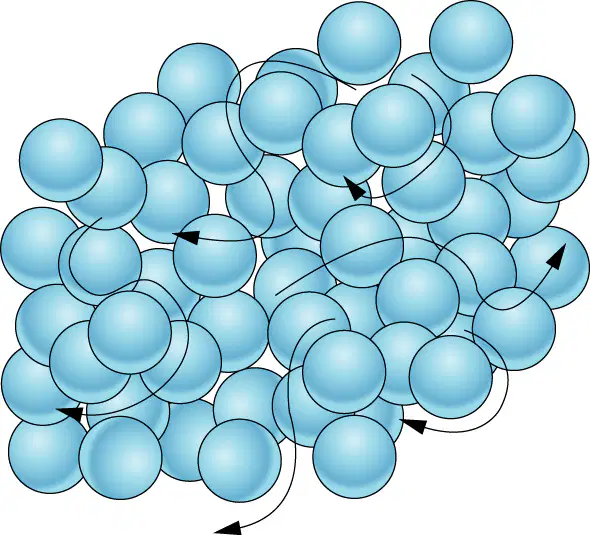
Which state is shown.
gas
solid
liquid
plasma
6
Multiple Choice

Which part of the graph has no temperature change
The part labeled ice
The part labeled freezing/melting
The part labeled water
The part labeled gas
7
Multiple Choice
Most matter __________ when heated
condenses
solidifies
contracts
expands
8
Multiple Choice
What occurs when the vapor pressure of a liquid equals the atmospheric pressure?
condensation
freezing
melting
boiling
9
Multiple Choice
What happens to KE when the temperature increases?
Nothing
It increases because particles move faster
It decreases because particles slow down
10
Multiple Choice
Why does the balloon shrink in liquid nitrogen?
The particles move apart and speed up
The particles slow down and move closer together since the temperature decreases.
Balloon shrink in all liquids
The liquid nitrogen damages the balloon.
11
Multiple Choice
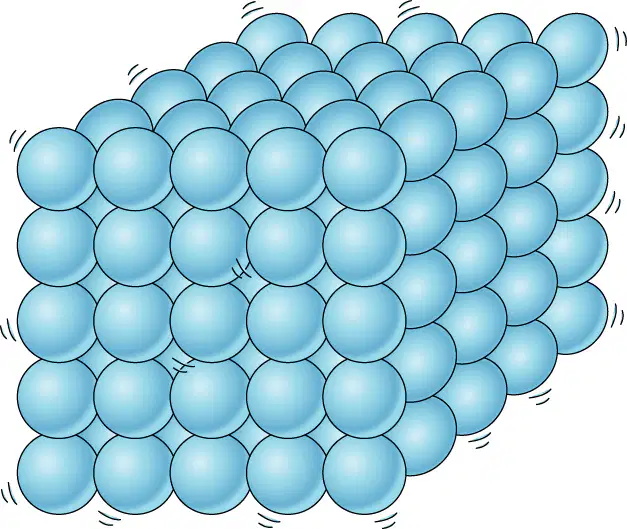
Which state of matter is shown?
gas
liquid
solid
plasma
12
Multiple Choice
This state has the lowest amount of energy. The particles mainly vibrate.
gas
solid
liquid
plasma
13
Multiple Choice
According to the kinetic molecular theory, all collisions between gas molecules are inelastic; all kinetic energy is lost.
True
False
14
Multiple Choice
Liquid changing to a gas just at its surface.
condensation
evaporation
melting
boiling
15
Multiple Select
What is true about an amorphous solid?
It does not have a melting point- it has a range of temperatures at which it melts
It behaves like all other solids
Glass and butter are examples
16
Multiple Choice
What is sublimation?
changing from a solid to a vapor
vapor changes to a solid
vapor changes to a liquid
liquid changes to a solid
Which best describes a solid?
The state of matter that has a fixed shape and fixed volume
The molecules of a solid move with a high speed in random order
The state of matter that has a fixed volume and takes the shape of its container
Molecules move past one another, vibrate, and rotate
Show answer
Auto Play
Slide 1 / 16
MULTIPLE CHOICE
Similar Resources on Wayground

12 questions
Human Biotechnology
Presentation
•
8th Grade

10 questions
Ch. 14.2 - Work and Machines
Presentation
•
9th Grade

13 questions
Big Bang Theory
Presentation
•
8th Grade

12 questions
Ch. 26.2 - Stars
Presentation
•
9th Grade

13 questions
Aquatic Ecosystems
Presentation
•
9th Grade

10 questions
Cellular Respiration
Presentation
•
9th - 10th Grade

13 questions
Speed v. Velocity
Presentation
•
8th Grade

15 questions
Building Pangea
Presentation
•
9th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Science

20 questions
MCAS Biology Review
Quiz
•
9th - 10th Grade

21 questions
Explore DNA Processes and Protein Formation
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

50 questions
living environment regents review
Quiz
•
9th Grade

17 questions
Unit 9 - Chemical and Nuclear Reactions
Quiz
•
9th - 12th Grade

50 questions
chem final review
Quiz
•
9th Grade

10 questions
Exploring Ecological Levels: Organism to Ecosystem
Interactive video
•
6th - 10th Grade

233 questions
BioPhysics B - Final Exam
Quiz
•
9th - 12th Grade